Question Analysis
This question involves VSEPR theory. The main focus is on applying the theory to determine the molecular geometry of a given molecule.
Key Concept Explanation
According to VSEPR theory, electron pairs around a central atom repel each other and arrange themselves as far apart as possible to minimize repulsion.
The molecular geometry depends on: 1. The number of electron domains (bonding + lone pairs). 2. Whether lone pairs are present.
Step-by-step Solution
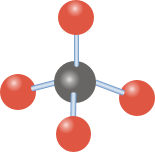
1. Structure analysis: Carbon has 4 valence electrons and forms 4 single bonds with chlorine atoms, using all its valence electrons. This leaves 0 lone pairs on the central carbon ...